Although light is generally said to be a wave, unlike the waves that occur at the surface of a body of water, it does not require a medium. As shown below light consists of an electric field and a magnetic field that intersect each other at a right angle as they move through a vacuum. The energy in light travels because of local fluctuating changes in electrical and magnetic fields; hence “electromagnetic” radiation.
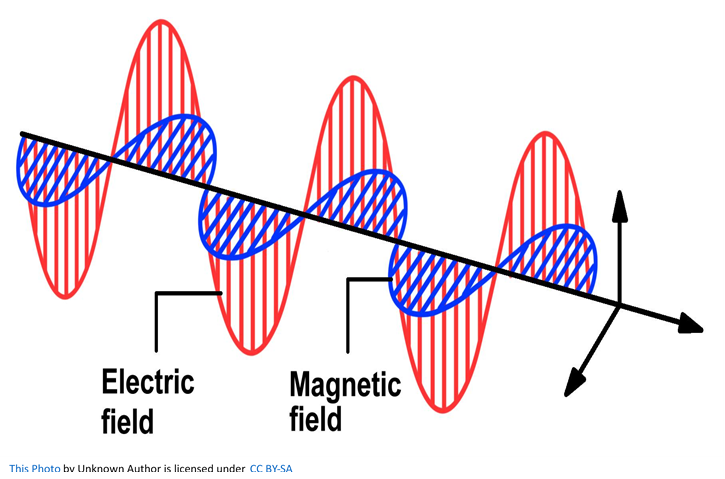
Let look at some of the important characteristics of light in general. Any wave is essentially just a way of shifting energy from one place to another, whether the transfer of energy is in waves on the sea or in the much more difficult to imagine electromagnetic waves in light. With waves on water, the energy is transferred by the bulk movement of water molecules; however, an individual water molecule doesn’t travel all the way across the Atlantic, or even all the way across a pond. Depending on the depth of the water, waves follow a roughly circular path from the point of origin. As they move up to the top of the circle, the wave builds to a crest; as they move down again, you get a trough. The energy is transferred by relatively small local movements in the medium. With water waves it is easy to draw diagrams to show this happening with real molecules. With light it is more difficult.
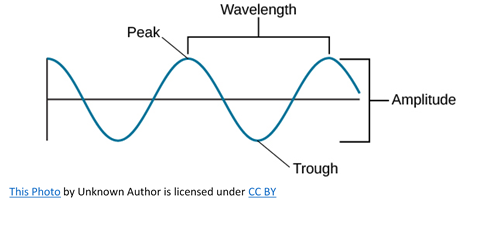
If you draw a beam of light in the form of a wave, without worrying too much about what exactly is causing the wave, the distance between two crests is called the wavelength of the light. It could equally well be the distance between two troughs or any other two identical positions on the wave. You must picture these wave crests as moving from left to right. If you counted the number of crests passing a point per second, you have the frequency of the light. It is measured in what used to be called “cycles per second”, but is now called Hertz, Hz. Cycles per second and Hertz mean the same thing. Light has a constant speed through a given substance. For example, it always travels at a speed of approximately 3 x 108 meters per second in a vacuum. This is the speed that all electromagnetic radiation travels not just visible light. There is a simple relationship between the wavelength, frequency, and the speed of light, shown below.
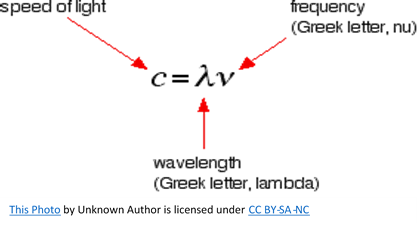
This relationship means that if you increase the frequency, you must decrease the wavelength and, of course, the opposite is true. If the wavelength is longer, the frequency is lower. It is important that you feel comfortable with the relationship between frequency and wavelength. Each frequency of light has an energy associated with it. The higher the frequency, the higher the energy of the light. Light which has wavelengths of around 380 nm to 435 nm is seen as a sequence of violet color. Various red colors have wavelengths around 625 nm to 740 nm. Which has the highest energy? The light with the highest energy will be the one with the highest frequency, that will be the one with the smallest wavelength.

Light consists of certain types of electromagnetic waves. Electromagnetic waves are referred to by different names in accordance with their wavelength, as shown here “Light” usually refers to electromagnetic waves in the range spanning infrared radiation and ultraviolet radiation, but in some cases, it refers only to visible light. Light with wavelengths in the range of approximately 380 nm to 780 nm is referred to as “visible light” and is the light that we humans can see with the naked eye. For example, light with a wavelength of 470 nm is blue, light with a wavelength of 540 nm is green, and light with a wavelength of 650 nm is red. Visible light could be described as the kind of light that we humans are familiar with because of our ability to see it.
As seen below visible light is a small subset of the entire electromagnetic spectrum.

For UV/Vis/NIR spectroscopy purposes the wavelength ranges are:
The ultraviolet region from 170 nm to 380 nm
The visible region from 380 nm to 780 nm
The near infrared region from 780 nm to 3300 nm
A nanometer (nm) is 10-9 meter. Most UV/Vis spectrophotometers are instruments that cover the 190 nm to 900 nm range when configured with a PMT detector or the 190 nm to 1100 nm range when configured with a diode detector. Some UV/Vis instruments can go into the lower NIR range with a PMT/InGaAs detector combination to cover the range of 190 nm to 1400/1600 nm. Typical UV/Vis/NIR instruments cover the 175 nm to 3300 nm wavelength range with PMT and InGaAs/PbS detectors.
People are very familiar with the visible light region, since these are the wavelengths that the human eye can see. The diagram above shows an approximation to the spectrum of visible light. The color order in the visible region is easy to remember using the mnemonic Roy G Biv. So, from long wavelength to short, the colors are red, orange, yellow, green, blue, indigo, violet. Now we must place the visible light region into the rest of the electromagnetic spectrum. This electromagnetic spectrum doesn’t stop with the colors you can see. It is perfectly possible to have wavelengths shorter than violet light or longer than red light. The figure above shows ultraviolet and the infrared regions that continue into x- rays and radio waves, amongst others.
Final thoughts – Don’t worry too much about the exact boundaries between the various sorts of electromagnetic radiation, because there are no boundaries. Just as with visible light, one sort of radiation merges into the next. Just be aware of the general pattern. Also be aware that the energy associated with the various kinds of radiation increases as the frequency increases (or wavelength decreases).
